Progressive supranuclear palsy (PSP) is a rare but serious neurodegenerative brain disorder that significantly affects a person’s movement, balance, vision, and cognitive functions. Symptoms often begin subtly with sudden falls, stiffness in the neck or upper body, or difficulty moving eyes up and down. Over time, PSP leads to increasing difficulty walking, speaking, swallowing, and performing everyday tasks.
PSP is classified as a tauopathy, meaning it is characterized by the abnormal buildup of the tau protein in certain brain cells. When tau clumps together, it damages and eventually kills these cells, leading to the hallmark symptoms of PSP.
There is currently no cure, but the field is advancing rapidly, and new treatments are now being tested to slow or stop the disease.
Logistical Hurdles in PSP Research
The low prevalence of PSP presents significant challenges for research and clinical trials. As a classified orphan disease, PSP has a limited patient population, making it difficult to recruit enough participants for large clinical trials. A key challenge is identifying and enrolling patients early in the disease, when treatments are most likely to be effective. Patients in more advanced stages of PSP may have too much neurodegeneration to respond, making it harder for trials to demonstrate meaningful results. To help overcome this, initiatives like the FTD Disorders Registry, an online database that collects information from people living with PSP and related conditions, allows researchers to more easily identify participants for clinical trials and biomarker research. Additionally, the CurePSP Centers of Care serve as both clinical hubs and active research sites, connecting patients to the latest clinical trials and research studies.
The Challenge of Measurement: The PSP Rating Scale
One of the biggest challenges in clinical research is figuring out how best to measure a treatment response. Clinical trials rely on biomarkers and patient assessments to track disease progression and determine the effectiveness of a drug. For PSP, the primary clinical assessment tool used is the PSP Rating Scale (PSPRS).
How the PSPRS Works
The PSPRS is a comprehensive questionnaire and observational tool designed to assess the severity of disability and impairment in patients with PSP. It was created to provide quantitative measures of disability across multiple domains of clinical impairment.
The PSPRS contains 28 items grouped into six functional categories:
- Daily activities (e.g., feeding, dressing, bathing, hygiene)
- Behavior (e.g., apathy, emotional lability, and disinhibition)
- Bulbar (e.g., speech and swallowing)
- Ocular motor (e.g., eye movement)
- Limb motor (e.g., slowness and rigidity in the arms and legs)
- Gait/midline (e.g., balance, postural instability, neck rigidity, and falls)
Through a mix of interview questions, observational tests, physical examination, and functional assessments, a clinician assigns scores across categories that combine into a total score ranging from 0 to 100. Higher scores indicate more severe symptoms, and changes over time can reflect disease progression or response to treatment. Because the scale covers multiple domains, it provides a comprehensive picture of how the disease is affecting a person’s functioning. Overall, the PSPRS score has demonstrated good inter-rater reliability and offers a standardized, widely accepted method for tracking PSP progression across different clinics and studies.
Challenges and Limitations:
While the PSPRS is a good scale for clinicians to measure disease progression, there are limitations for its use as a lone primary endpoint in clinical trials. It requires specialized training for consistent and reliable administration, especially for the exam-based items. Researchers also question whether every item on the original scale reflects changes that matter the most to patients – some items may carry more real-world significance than others. In addition, certain items on the scale change very slowly over time or plateau as the disease advances, making it challenging to detect meaningful differences between treatment and placebo groups. The scale is also heavily weighted toward motor symptoms, which means cognitive and behavioral changes may not be fully captured.
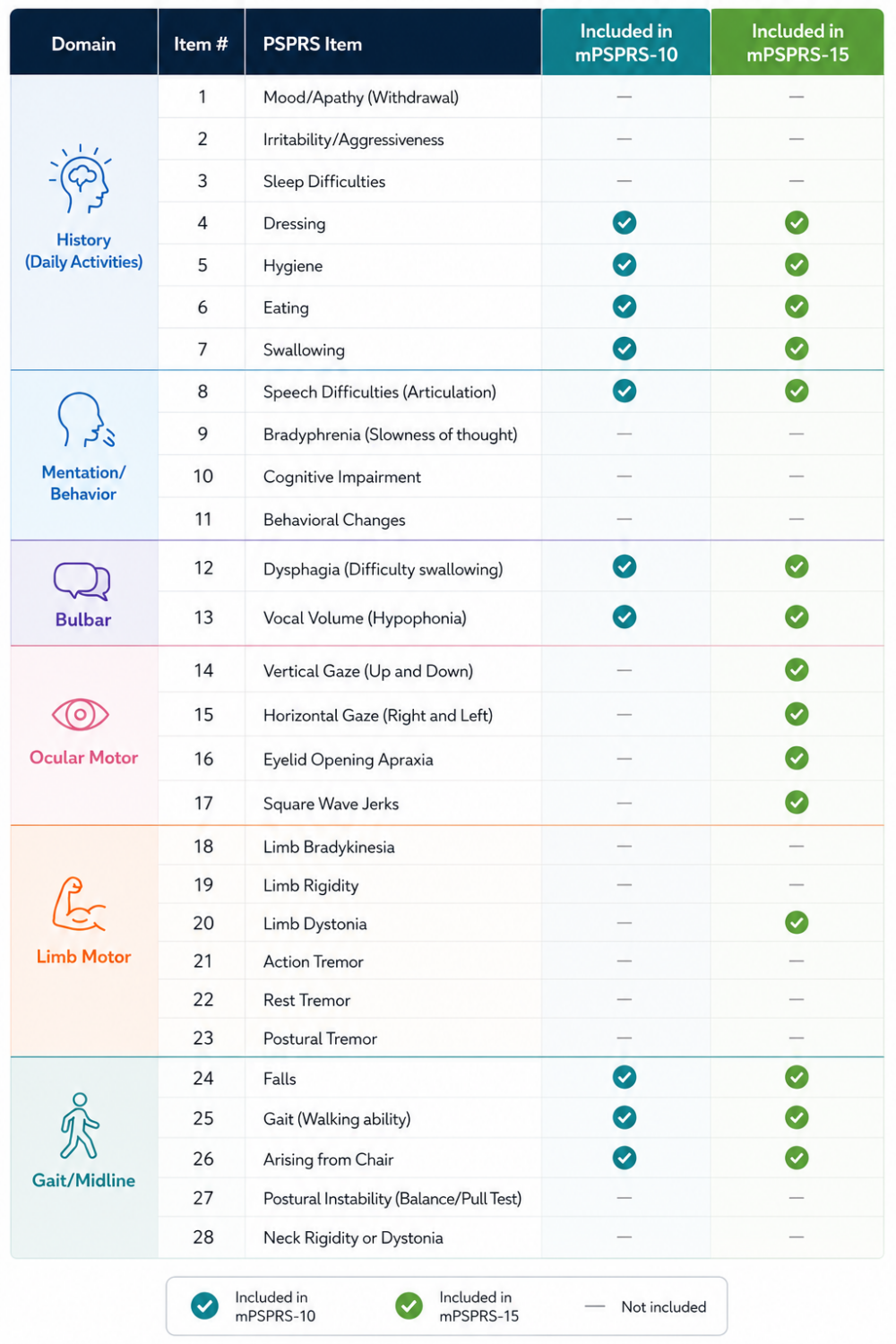
To address these concerns, modified versions of the PSPRS have been developed:
- The 10-item modified subscale (mPSPRS-10) originated from guidance by the U.S. Food and Drug Administration (FDA). It focuses on daily activities, bulbar, and gait/midline items, emphasizing core functional decline deemed critical for assessing treatment effects in clinical trials.
- A 15-item modified subscale (mPSPRS-15) includes the 10 items from mPSPRS-10 with additional measures related to eye movement and muscle control. It has been shown to be more sensitive than the full 28-item scale in detecting meaningful changes in disease progression, making it a strong candidate for future primary endpoints in clinical trials.
- Other modifications of the scale include ongoing efforts to create subscales that better capture cognitive and behavioral decline (such as apathy or executive dysfunction), which significantly impact quality of life but are not fully represented by the motor-focused scales.
The Future: Toward Objective Measures and Biomarkers
While the modified PSPRS subscales represent a significant improvement, the field recognizes that a drug’s success should not rely solely on clinical assessment scales that may have elements that are subject to interpretation. A major hurdle is the lack of an objective test, such as a blood or neuroimaging test, that can definitively demonstrate how well a drug is working for patients. Researchers are now pushing to identify biomarkers that can track disease progression or response to treatment. Validating these biomarkers is not only crucial to determine the efficacy of a drug but would also allow for clinical trials to be run more efficiently.
To learn more about participating in research, individuals and families affected by PSP can explore opportunities through the FTD Disorders Registry. For those navigating the disease, CurePSP provides connections to specialized care, clinical studies, and patient advocacy support. Finally, researchers can explore additional tools and resources available here.

